
Secondary outcomes will include measures of the durability and effects of the iRPE patch.Īge-related macular degeneration (AMD) is a leading cause of vision loss among people 55 years old and older. The primary outcome is the safety of iRPE transplantation. If adequate safety is demonstrated among these participants, up to an additional seven eligible participants with best-corrected visual acuity between 20/80 and 20/500 in the worse-seeing eye will be enrolled. Five participants meeting eligibility criteria, including best-corrected visual acuity between 20/100 and 20/500 in the worse-seeing eye being considered for the study, will be enrolled initially. The goal is to transplant the iRPE patch into an area alongside a border of geographic atrophy causing central vision loss. The iRPE patch consists of an approximately 2 X 4 mm oval sheet in which cells are grown on a biodegradable PLGA scaffold material. In this investigational, first-in-human, phase I/IIa, single-arm clinical trial, participants will undergo subretinal transplantation of autologous induced pluripotent stem cell-derived retinal pigment epithelium (iRPE) in one eye and will be followed for five years after surgery. It does not store any personal data.Official Title: A Phase I/IIa Trial for Autologous Transplantation of Induced Pluripotent Stem Cell-Derived Retinal Pigment Epithelium for Geographic Atrophy Associated with Age-Related Macular Degeneration The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Depending on the type of corneal transplant you have and how you heal, it may take up to a year to fully recover from this surgery. Pain medication may be taken as necessary. As you recover from surgery, you will need to use prescribed eye drops, wear protective eyewear such as sunglasses and be wary not to rub your eye or eyes. 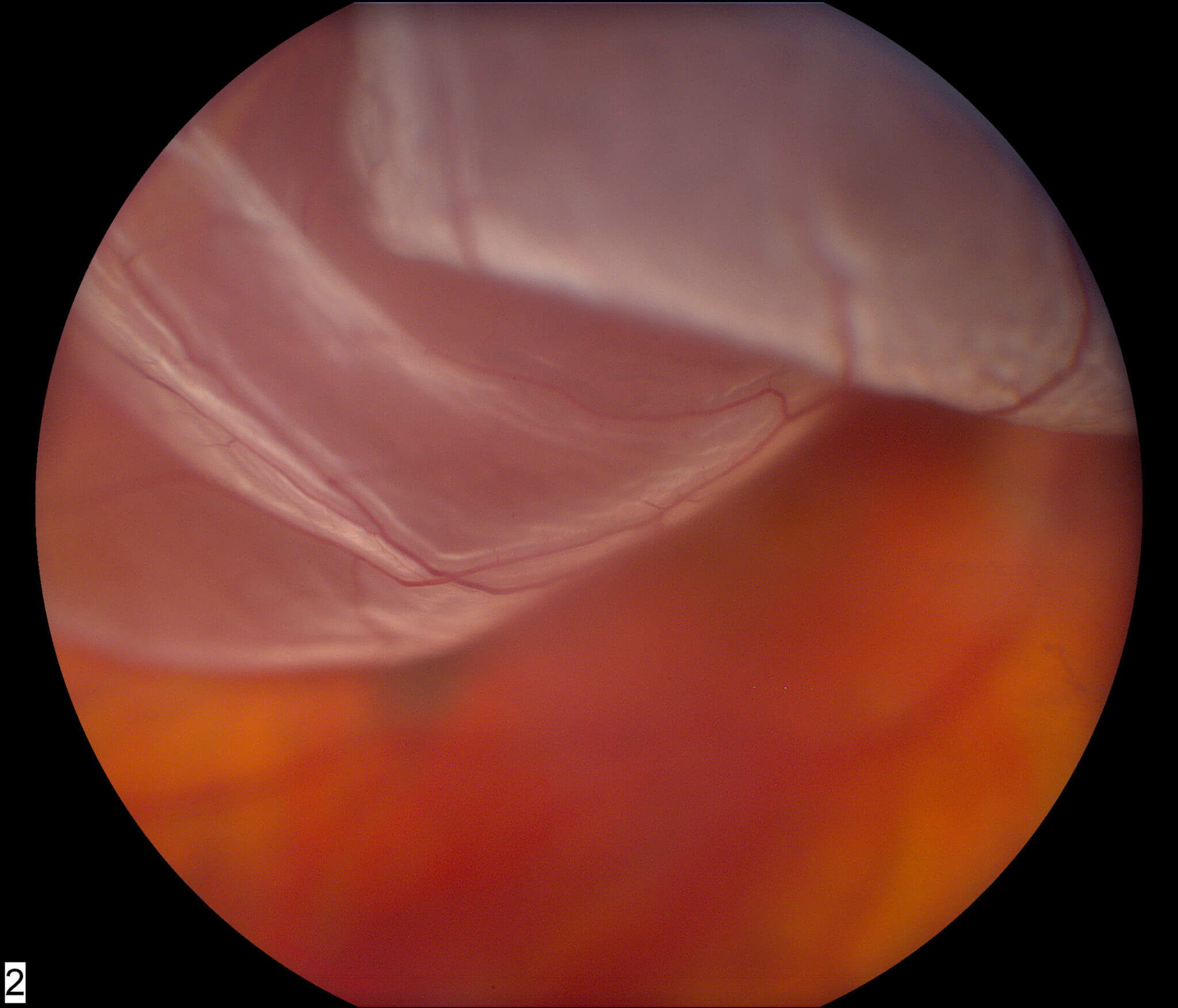
Keep in mind that you will not be able to drive after corneal transplant surgery and will thus need to make arrangements for someone to drive you home after your surgery. Common reasons for corneal transplant surgery are keratoconus, corneal scarring and endothelial dystrophy. Dr Enslin Uys will refer you to one of his colleagues that perform this surgery on a regular basis. If only specific layers of your cornea are damaged, a lamellar corneal transplant may be recommended as it is less invasive, and the recovery time is generally much faster than that of a full thickness corneal transplant. During a full thickness corneal transplant (penetrating keratoplasty), the ophthalmologist will transfer all layers of the cornea from a donor to restore your sight.ĭuring a lamellar corneal transplant (endothelial keratoplasty) on the other hand, only selected layers are transplanted. 
There are a number of cornea replacement surgeries which can be performed, one being a full thickness corneal transplant and the other, a lamellar corneal transplant. Corneal transplant surgery, or keratoplasty, involves the removal of part of the cornea so that it can be replaced with a new section taken from a donor.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
